EXTEDO on X: "“Moving Forward with #IDMP” - only one day left to join our complimentary webinar on 21 March 2024 at 16:00 CET. Registration: https://t.co/f1voHP3NHh Not sure if you can make

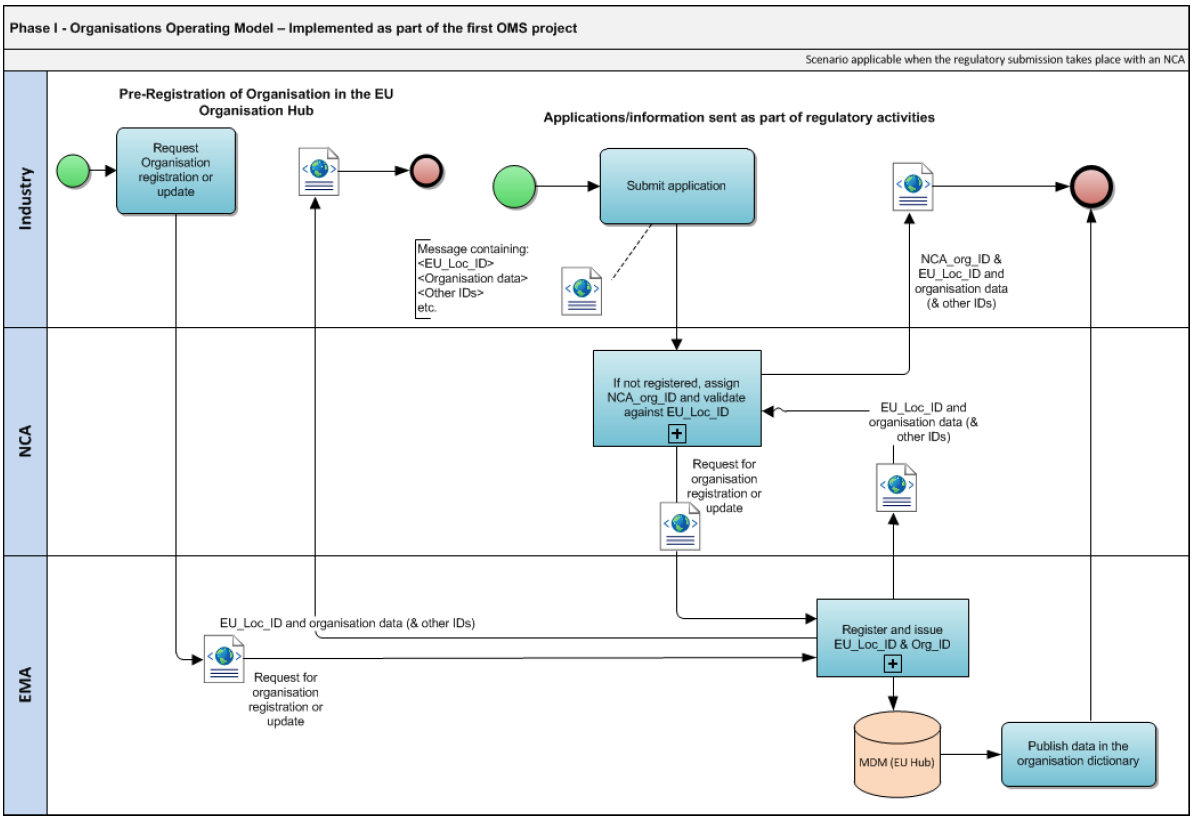
Preparing for ISO IDMP — Not as Black as It's Painted (When You Know How to Do It) - Striped Giraffe